Application of New Paper-based Microfluidic Sensing Materials in Food Analysis
-
摘要: 相较于其他食品分析检测技术成本高和制作方法复杂等缺点,纸基微流控技术具有制作成本低,制造工艺简单和无毒环保等优点,已在众多领域广泛应用。本文简要介绍了纸基微流控技术,重点讲述了有机分子或化合物材料、纳米材料和聚合物材料在内的探针的传感性能及其检测极限,有望帮助读者更好地理解μPADs中使用的各种化学和生物探针的传感机制,并促进其在该领域的进步。纸基微流控新型传感材料和纸基微流控技术在农药残留、致病菌、重金属和添加剂等食品分析中的检测机理以及其在整个检测体系中发挥简便快捷等优势的同时得到理想的实验结果,为其在快检领域的发展提供基础,并对该技术的未来发展方向和应用前景进行了展望。Abstract: New paper-based microfluidic technology has been widely used in many fields due to their advantages such as easy preparation, low cost, and non-toxic environmental protection, compared with the shortcomings of other food analysis and detection techniques such as complex production method and high cost of preparation. The outline of new paper-based microfluidic technology is briefly summarized and the probes including organic molecules, nanomaterials and polymers their structural activity with regard to sensing performances along with their limit of detection are also discussed. This review is expected to assist readers for better understanding of the sensing mechanisms of various chemo and bio-probes utilized in µPADs, as well as promote their advancement in the field. The detection mechanism and its advantages in the whole detection system at the same time to get the ideal experimental results of new paper-based microfluidic technology in the food analysis of pesticide residues, pathogenic bacteria, additives, heavy metals and other substances are mainly introduced. This study would provide the theoretical basis and references for fast detection field. The future challenges and application prospects of this technology are prospected.
-
Keywords:
- paper-based microfluidic /
- sensor material /
- detection techniques /
- food analysis
-
食品安全问题事关民生国计,已经引起社会各界的广泛关注[1]。现阶段,常用于食品分析检测技术的方法主要有电位分析法[2]、气相色谱法[3]和高效液相色谱法[4]等,这些方法具有灵敏度高、选择性好、分析速度快和检出限量可降低等优点,适用于食品中微量成分的测定[5]。然而,在实际应用中存在昂贵、低效、样品制备困难和实验仪器操作复杂等缺点[6]。研究发现,侧流免疫层析法[7]、传感器法[8]和酶抑制法[9]等方法具备操作简单、检测迅速和成本效益高等优势,适用于现场快速检测,但灵敏度较差、不稳定、不易携带和难以大规模生产等缺点限制了其应用范围[10]。随着检测技术进一步发展,微流控纸基分析装置(microfluidics paper-based device,µPADs),即微流控纸芯片的出现,为食品分析领域提供了一种集成化、便携化和低成本化的检测方法[11-12]。作为一种终端分析工具,μPADs由于样品制备和信号放大均在设备外完成,从而无法满足复杂的分析需求。因此,需开发性能优异的μPADs,如将各种分子探针功能化并引入纸芯片中,利用纳米材料[13]、生物分子[14]和其他几种探针构建新型μPADs传感器,提高其在分析检测过程中的灵敏度、选择性、稳定性、重现性及准确性。基于上述优势,该技术已成为对各类食品成分定性定量的主要分析检测方法。本文通过介绍μPADs新型传感材料及其在食品分析中的应用,旨在加深读者对µPADs中各种化学和生物探针传感机制的理解,为纸基微流控技术进一步的优化设计提供理论依据。
1. 纸基微流控新型传感材料
纸芯片在不同领域中展现诸多优势,在分析稳定性和检测灵敏度等方面仍有很大的进步空间。因此,研究人员提出了一种策略,将各种探针材料与纸芯片互相结合,如有机分子和化合物、纳米材料和聚合物材料与纸芯片结合[15]。通过修饰各种官能团改善纸芯片的性质或利用纳米材料独特的光学性质、表面易功能化及良好的生物相容性修饰纸芯片。此外,根据μPADs的需要合成聚合物材料对纸芯片进行修饰[16]。将此类材料作为传感基质,实现了纸芯片在检测过程中灵敏度、选择性和准确性等方面的突破。
1.1 有机分子或化合物材料
格里斯试剂(Griess)作为一种有效的分子探针,常用于检测食品和环境样本中的亚硝酸盐。Arvand等[17]研制了一种用于实际样品中亚硝酸盐含量测定的比色微流控纸基分析装置。亚硝酸盐使Griess试剂中对氨基苯磺酸重氮化,生成重氮苯磺酸,显示出玫红色,但反应过程中Griess试剂易氧化分解,酒石酸可提升Griess试剂的稳定性。通过酒石酸改良Griess试剂作为探针材料,基于改良后Griess分析的比色法应用于亚硝酸盐的测定,其中,检测限(limit of detection,LOD)为15 µmol/L。对实际样品自来水进行回收实验,回收率为96%~107%,相对标准偏差(relative standard deviation,RSD)为0.78%,说明该体系有较高的灵敏度、回收率和精确度,可以实现对实际样品中亚硝酸盐的检测。该方案表明有机分子通过合适的改良可提高自身化学性质,为探针材料的优化设计提供了新思路。
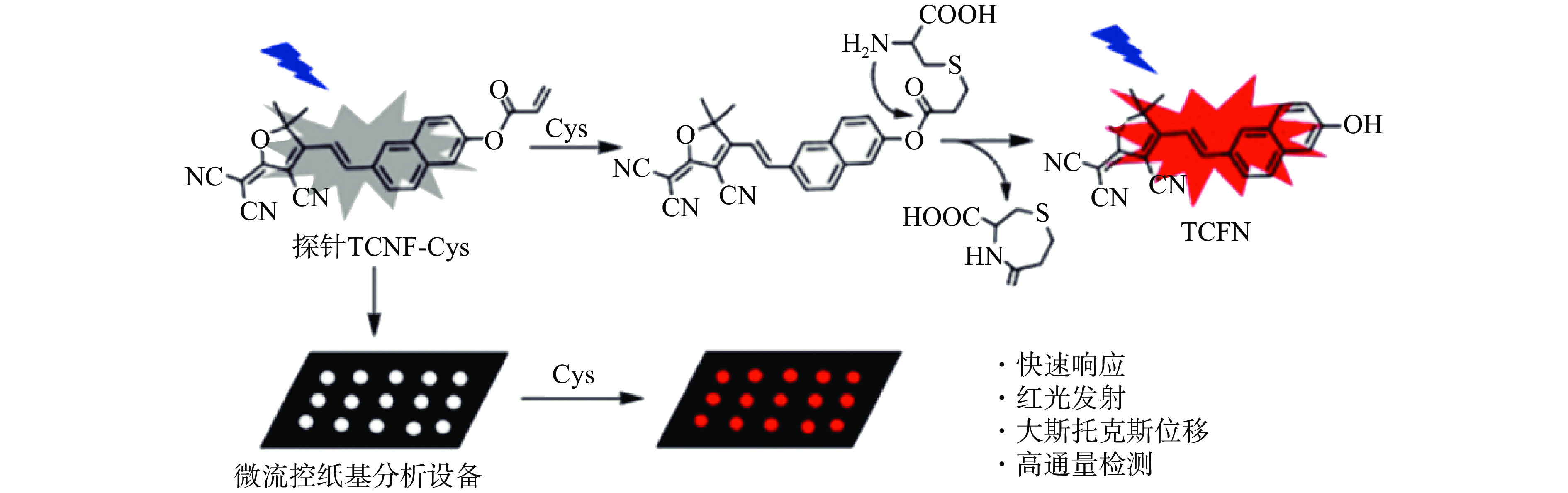
纸芯片与荧光相结合可用于氨基酸的检测。Zhou等[18]利用2-(3-氰基-4,5,5-三甲基呋喃-2(5H)-亚基)丙二腈(2-(3-Cyano-4,5,5-trimethylfuran-2(5H)-ylidene)malononitrile,TCF)与6-羟基-2-萘甲醛缩合,制备TCFN-Cys探针并引入纸芯片中进行半胱氨酸(Cysteine,Cys)的检测(图1)。该探针以萘衍生物作为发色团,丙烯酸酯作为淬灭和识别位点。发色团中强大的分子内电荷转移可以实现红色荧光和较大的斯托克斯位移。丙烯酸酯基团可以与Cys反应生成具有增强红色荧光的染料TCFN。研究结果表明,其中半胱氨酸于0~5 mmol/L的浓度范围在纸装置上的LOD为12.4 nmol/L,说明该方法适用于半胱氨酸的高灵敏快速检测。Zhang等[19]开发了一种利用荧光探针对Cys进行比色检测的微流控纸基分析装置。将间苯二酚与7-硝基苯并呋喃嗪结合,合成用于Cys比色检测的荧光探针。在探针与Cys反应后,间苯二酚被释放,纸芯片颜色由黄色变为红色。Cys浓度线性范围为0.04~70.04 μmol/L,LOD为16 nmol/L。鉴于此,该方案具有可移植性、可处置性及灵敏度高等优点。通过将荧光技术和μPADs相互结合,为食品中各种成分进行高通量和高选择性分析提供了一个新的平台,也为μPADs的发展提供了一个新的思路,将其与不同技术相结合,既简化了传统技术的操作步骤,又拓宽了μPADs的检测范围。
通过青霉胺(Penicillamine,DPA)热解柠檬酸合成石墨烯量子点(Graphene Quantum Dots,GQDs)从而制备GQDs-DPA探针,进而利用一种创新的微流控纸基分析装置对金属离子进行检测[20]。GQD-DPA可在纸基传感器上选择性识别Cu2+。该传感探针在0.001~5 ppm的浓度范围内呈现良好的线性关系,在实际样本中的定量下限为0.1 ppm。该方法实现了Cu2+的可视化检测,具有环保、经济和准确的优势。此外,氨基酸的加入可增强探针的识别效果,提高传感系统的准确性及检测其他离子,如在含有巯基的Cys存在的情况下,可以检测到Fe3+和Hg2+。基于微流控纸芯片上选择性检测不同金属离子的技术是完全创新的,这一发现为纸芯片选择性检测提供了新的研究方向。
1.2 纳米材料
纳米材料由于其具有优异的稳定性、低毒及良好的生物相容性,成为微流控纸芯片分子探针的热门选材,并在食品分析领域显示出非凡的应用潜力。
氧化锌纳米棒制备微流控纸分析装置可用于多种有机物检测。Feng等[21]以葡萄糖和尿酸为研究对象,利用双酶促反应在µPADs上进行比色检测。该方案中氧化锌纳米棒的均匀分布促进了酶的固定化,显著提高了比色信号且有效地克服了咖啡环效应。葡萄糖和尿酸的LOD分别为3 µmol/L和4 µmol/L,RSD小于1.57%。研究结果表明,新开发的µPADs具有良好的重复性和可靠性且与以往报道的µPADs相比,具有快速、无加热和灵敏度高等优点。此方案为葡萄糖和尿酸等生物分子检测提供了一种简单、低成本和便携式的测试平台。
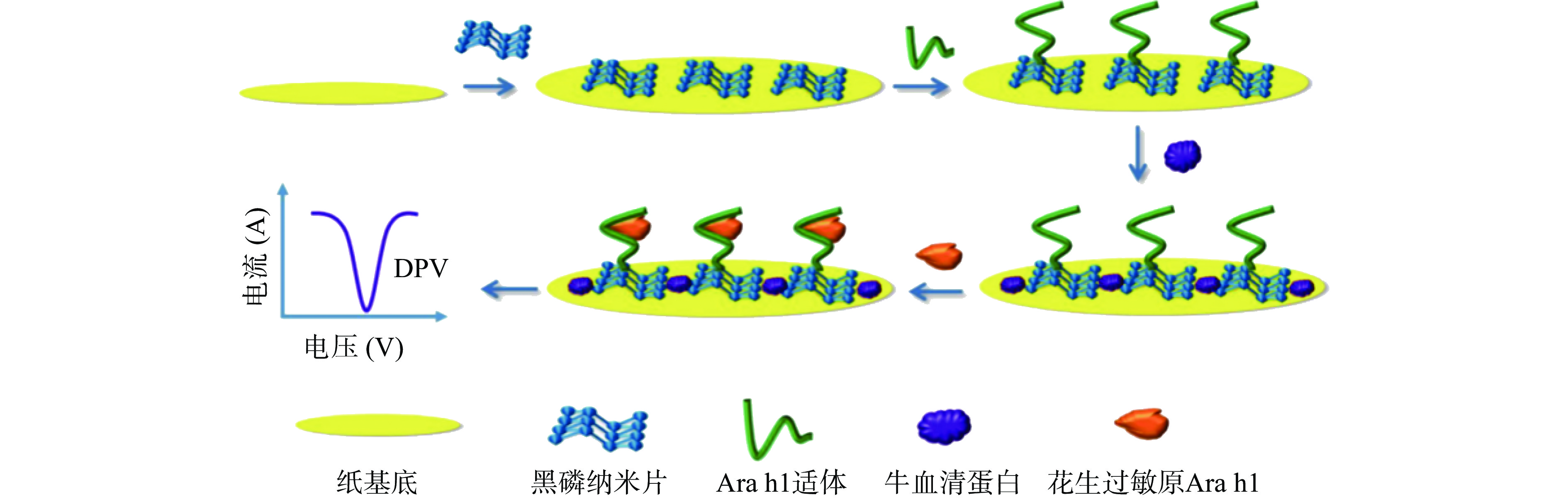
3D打印技术的成熟促进了三维纸芯片的研究。Jiang等[22]开发了一种三维纸基微流控电化学纳米传感器,用于花生过敏原Ara h1的快速检测。采用黑磷纳米片作为分子识别探针,可增强电化学检测的特异性和选择性。在传感过程中使用亚铁氰化物作为氧化还原探针,选取一种对Ara h1没有标记的适体,能够在20 min内实现无标记电化学检测(图2)。得出线性范围为50~1000 ng/mL,LOD为21.6 ng/mL。该适体传感器具有高特异性、高灵敏度及高选择性等优势,并已成功应用于饼干面团样品中Ara h1的检测且不需要复杂的实验室测试。
金纳米粒子因具有模拟酶特性常用于食品和药物检测等方面。Shariati等[23]提供了一种在微流控纸基分析装置上利用功能化的AuNPs对测定金属离子进行测定的方法,该方法使用N,N′-二(2-羟乙基)二硫氧酰胺修饰金纳米粒子并对Hg2+进行可视化检测。Hg2+的浓度与改性AuNPs的颜色之间呈现出良好的线性关系,其线性浓度的范围为0.025~2.0 µmol/L,LOD为15 nmol/L。该方法具有可处理性强、快速简便、便携性好、成本低和使用材料少等优点,适用于食品和药品等样品中Hg2+的测定。Pinheiro等[24]利用AuNPs制备检测葡萄糖、尿酸和游离胆固醇的微流控纸基分析装置。葡萄糖可通过原位AuNPs合成法开发的µPADs进行半定量测定,尿酸和胆固醇利用改性的AuNPs根据光学性质的变化可进行定量测定。该实验结果表明,利用不同的物质或方法对AuNPs进行修饰,可以使AuNPs具有专一性,因此可根据不同需求对纳米探针进行针对性修饰从而实现专一性检测。
1.3 聚合物材料
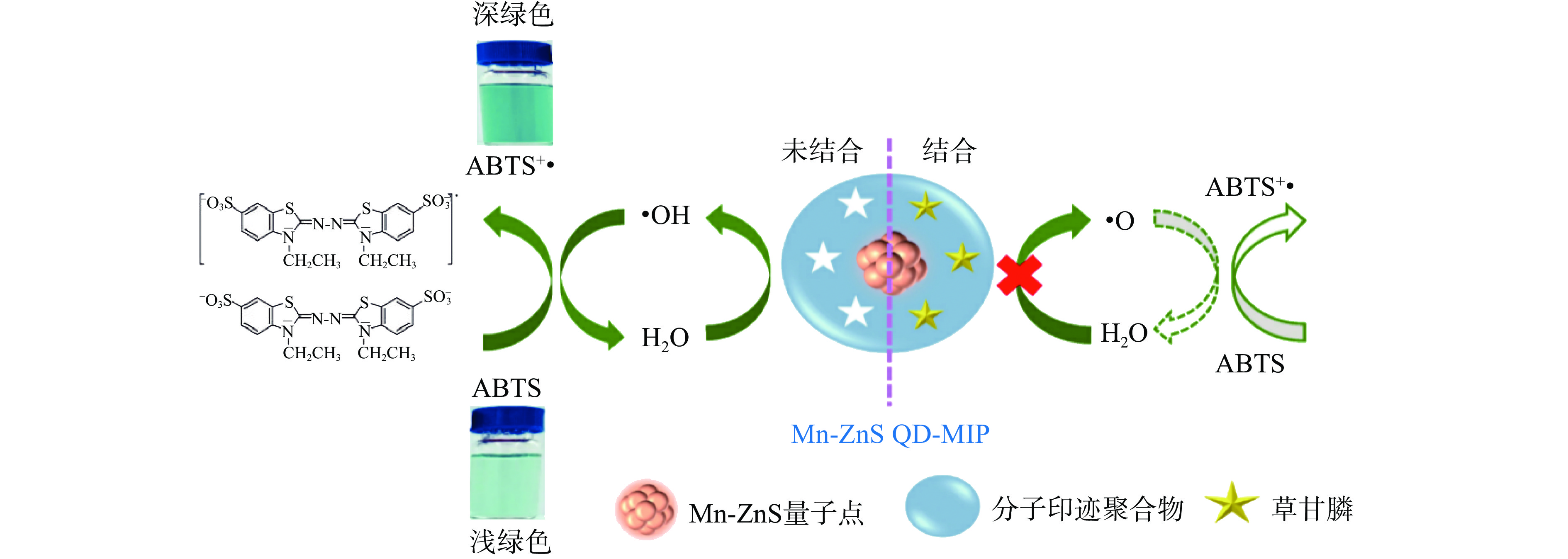
聚合物作为最后一类修饰µPADs的材料,可用于农药或重金属等检测。Sawetwong等[25]报道了一种新型的三维微流控纸基分析装置(3D-µPADs),其在Mn-ZnS量子点中嵌入分子印迹聚合物Mn-ZnS QD-MIP制备了3D-µPADs,并用于全谷物样品中草甘膦的比色检测。其中Mn-ZnS QD-MIP作为2,2′-联氮-双-3-乙基苯并噻唑啉-6-磺酸(Agreement of Basic Telecommunications Services,ABTS)与H2O2反应的竞争抑制剂。草甘膦选择性嵌入Mn-ZnS量子点表面的空腔,抑制ABTS氧化使其颜色变为浅绿色(图3),根据反应颜色的变化确定草甘膦的浓度。在0.005~50 μg/mL的工作范围内测定草甘膦,LOD为0.002 μg/mL,即3D-μPADs显示出很高的精确度。此外,制作的3D-μPADs还具有良好的再现性。开发的3D-μPADs已成功应用于全谷物样品中草甘膦浓度的测定,并显示出较高的选择性和灵敏度且无复杂的仪器对食品质量控制和现场环境监测具有重要意义。
利用碳点修饰纸芯片可应用于生物检测。Ortiz-Gomez等[26]研制了一种基于集成碳点(Carbon Dots,CDs)的微流控纸基分析装置,用于生物硫醇(GSH、Cys和Hcy)的荧光检测。通过一锅法利用乙烯基砜(Vinyl sulfone,VS)将CDs固定在纸基上,生物硫醇可对CDs的蓝色荧光强度产生影响,从而可以确定生物硫醇的浓度。在GSH为1~200 μmol/L,Cys和Hcy为5~200 μmol/L检测范围中,得到GSH和Cys的LOD为0.3 μmol/L及Hcy的LOD为0.4 μmol/L。表明该方法具有高灵敏度和高选择性,将集成碳点的纸基微流控分析装置与荧光检测相结合,可拓展可视化检测的相关研究。
钙离子的量化依赖于离子载体掺杂的离子选择性纳米光电二极管修改的变色检测通道长度,不需任何仪器可直接观察,结合纳米光电二极管的µPADs是一种灵敏且可重复使用的工具,可用于无设备现场分析真实样品中的Ca2+的检测,检测范围在0.05~5 mmol/L,低于之前在任何微流控纸基分析装置中的检测范围[27]。因其低成本及制造方法简便,在实际应用中可大批量生产。但该方法中基于距离的纸基设备需要较长的分析时间来获取定量光电信号,如几厘米长的检测通道则需45 min才可获取结果。此外,基于距离的比色信号只能通过纸张中的纤维网络进行样品的传输。
基于上述内容,将三大类材料开发的各种探针进行简要讨论,说明了探针及其各自的分析物和检测限见表1。
表 1 纸基微流控不同传感材料性能对比研究Table 1. Performance comparison of different sensing materials for paper-based microfluidics探针材料 检测方法 检测物质 LOD 参考文献 格里斯试剂 纸芯片比色法 亚硝酸盐 15 µmol/L [17] 间苯二酚与7-硝基苯并呋喃嗪 纸芯片荧光法 半胱氨酸 16 nmol/L [19] ZnFe2O4磁性纳米粒子 纸芯片比色传感器 双酚A 6.18 nmol/L [28] 用抗体和辣根过氧化物酶修饰的纳米金-单链DNA 纸芯片比色传感器 病原体抗原 10 pg/mL [29] 溴甲酚绿试剂 智能手机三维纸芯片比色法 白蛋白 3.68 g/L [30] 氯酚红-β-D-半乳吡喃糖苷和磷酸品红 纸芯片比色法 金黄色葡萄球菌和大肠杆菌 106 CFU/mL [31] 2′-羟基查尔酮衍生物 纸芯片荧光法 白蛋白 16.4 nmol/L [32] 以二乙烯基砜作为同型二功能试剂的碳点 纸芯片荧光法 生物硫醇 0.3 μmol/L [26] 1,5-二苯基碳酰二肼修饰的纳米银 纸芯片比色法 半胱氨酸和同型半胱氨酸 0.16 μmol/L [33] 金纳米粒子适配体 智能手机纸芯片比色法 多氯联苯 70 ng/L [34] 碘化汞 纸芯片比色法 重金属汞 20 mg/L [35] 有机分子及化合物材料、纳米材料和聚合物材料修饰的纸芯片,可与其他方法结合构成完整的纸基微流控分析装置。比色法、荧光法和化学传感器等是最常用的方法,其中,比色法与被修饰过的纸芯片相结合,相较于其他方法,检测结果更直观,其对应的研究结果也更丰富。纸芯片与其他检测方法的结合,可简化检测过程,节约成本,但探针中若有易挥发、易分解成分则难以修饰纸芯片,达到理想的结果,有待进一步明确。此外,为进一步扩展纸芯片的应用范围,3D打印技术的发展,为其提供了新的方向,这类研究还处于萌芽状态,需进一步探究。
2. 纸基微流控在食品分析中的应用
食品安全问题已经严重威胁到人民的健康生活和经济的可持续发展。人们对食物过敏、食物中毒和食源性疾病相关问题的关注,促进了食品安全监管的演变及食品分析方法的发现[36-37]。现阶段,食品分析需要快速、准确、敏感和具有成本效益的方法来监测和保证安全和质量,以满足严格的食品立法和消费者的需求。酶抑制法[38]、光谱检测法[39]和色谱检测法[40]等方法存在设备复杂、检测成本高、自动化程度低、耗时和需要大量试剂等问题[41]。而微流控纸芯片的出现,巧妙地解决了上述问题,且该装置具有样品消耗少、检测快、操作简单、多功能集成、体积小、多功能检测和便携性等诸多优点[42],为食品分析领域快速检测奠定了坚实基础。
2.1 农药残留检测
农药残留会使人腹泻、呼吸困难和睡眠状况差。每年,因误食含有农药残留的食物而患癌的人数在以15%的速度增长[43-44],因此寻找快速检测农药残留的方法刻不容缓。
电化学检测方法是检测农药残留最常用的方法之一,但需要复杂的电极制造过程及高昂的检测成本。纸基微流控技术的发展,弥补了传统检测方法的不足。
无机农药是由无机矿物质制成的农药,药效强,易残留。Orouji等[45]利用未经修饰的银纳米粒子作为纳米探针,制作纸基微流控比色检测体系,在pH为4.5~9.5的范围内测定单磷和甲基叠氮磷农药,LOD分别为0.24和0.16 μmol/L。此方法为开发高灵敏度、选择性、便捷性和可视化农残检测器提供了一种有效的手段,但关于修饰纳米粒子特性的研究却很少。
有机农药由人工合成,药效挥发快。Deng等[46]介绍了一种新方法,用于快速分析有机磷农药残基中的三氯氟酮。结合半定量法,三氯氟酮的LOD为1.65 µg/mL。微流控纸质磷酸化检测芯片具有高灵敏度,适用于快速检测机磷农药。Nsibande等[47]用半胱氨酸修饰的CdSeTe/ZnS量子点,量子点通过缩合过程用三甲氧基硅烷官能化,在量子点表面引入可聚合乙烯基。将修饰后的量子点用作荧光传感探针,在纸芯片上进行阿特拉津的检测,得出LOD为0.8×10–7 μmol/L,极低的检测限表明该种方法具有较好的灵敏度。纸基微流控技术对于农药残留的检测,简化了以往复杂的检测过程,解决了检测设备携带不方便等问题,具有良好的特异性。
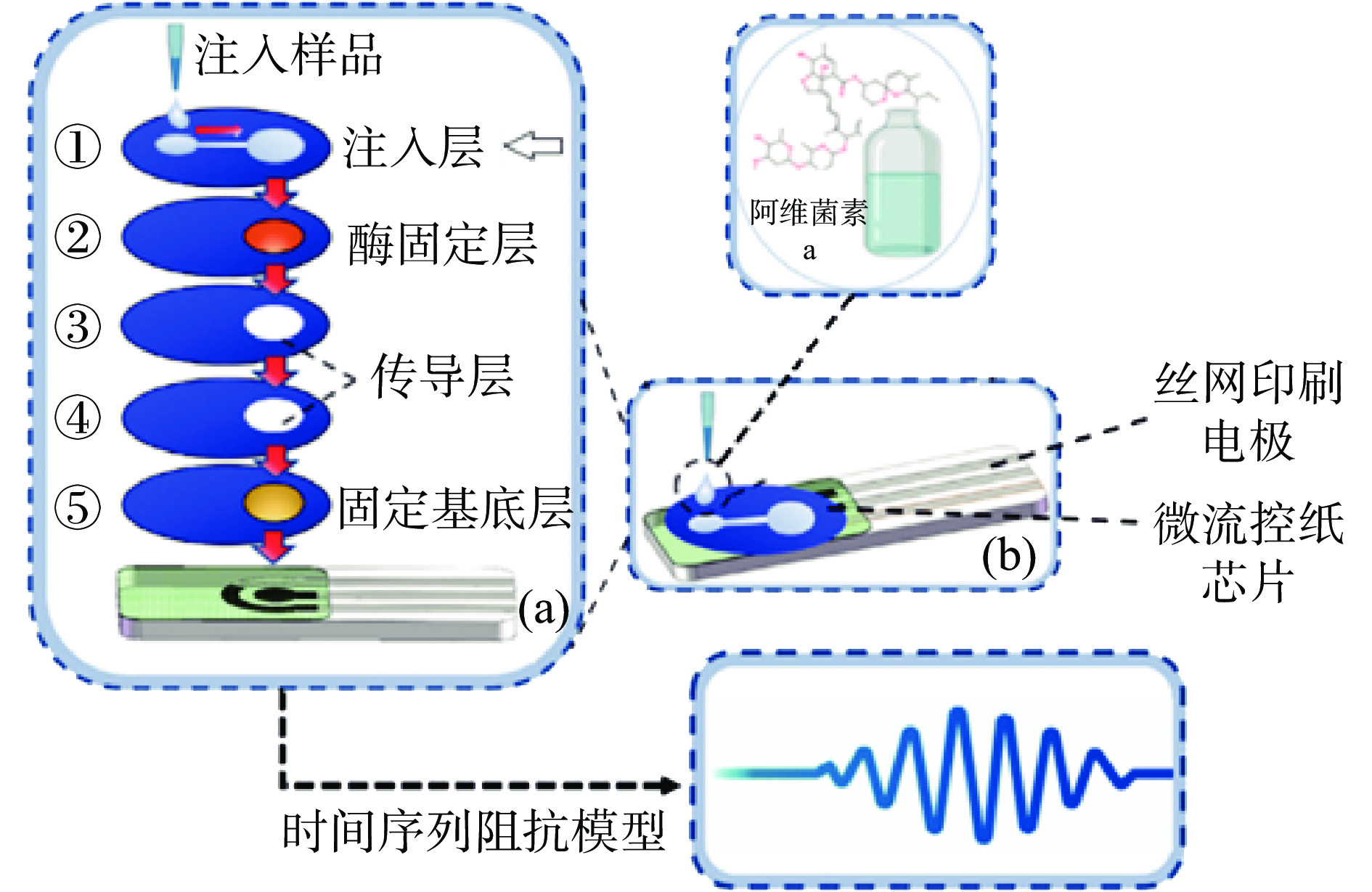
生物源农药是由植物、抗菌素和微生物等生物制成的农药,毒性较低。Yang等[48]提出了一种基于多层微流控纸芯片的农药残留鉴定方法。微流控纸芯片集成了酶抑制剂和无标记丝网印刷碳电极,选择阿维菌素作为研究对象,通过分析15 min内酶抑制剂引起阻抗数据的变化实现纸片上的检测(图4)。对实际样本进行检测,以生菜为例,分类准确率达到了93%左右。因此,该方法在快速鉴定不同农药方面具有良好的稳定性和特异性。目前,三氯氟乙烯检测的标准方法是气相色谱法。与气相色谱相比,微流控纸质磷酸化检测芯片可以在短时间内提供直观可靠的结果且具有更高的便携性,还可以针对不同农药进行同步检测。然而,该方法在检测过程中容易发生交叉污染。
2.2 致病菌检测
食品致病菌是食品安全最严重的威胁之一。人误食经致病菌污染的食物可能会出现呕吐、腹泻、肠道感染等症状,严重的可导致神经系统受损、致癌等。因此,寻找能够检测食品中致病菌的方法非常重要。在所有食源性疾病中,致病性最多的三种细菌是大肠杆菌、沙门氏菌和单核增生李斯特菌[49-50],食用被大肠杆菌O157:H7污染的食物可导致出血性结肠炎和出血性尿毒症综合征[51]。
传统的检测方法需要对细菌进行培养和富集,整体检测较长,然而微流控检测技术可以有效地解决这一难题。目前各种类型的微流控体系已用来对细菌性病原体进行检测。Karuppiah等[52]开发了一种丝网印刷纸基微流控生物传感器,用于检测食品中的大肠杆菌群,该生物传感器具有超高灵敏度和携带方便等优势。纸基微流控集成生物传感器,用于现场检测最常见的食源性病原体[53]。Park等[54]将基于微流控纸芯片的环介导等温放大技术与碳纳米管多层生物传感器相互结合并用于大肠杆菌O157:H7的检测。经抗体功能化的碳纳米管多层膜可以选择性地捕获、培养和释放细菌,减少了细菌培养的周期,缩短了整体的检测时长,利用与LAMP结合的微流控纸芯片对释放的细菌浓度进行分析。该传感平台能够检测到低至1 CFU/mL的浓度,这比以前报道的方法更敏感。该传感平台具有成本低、操作方便等优点,是食品安全中检测大肠杆菌的潜在平台。
2.3 重金属检测
食物中的重金属元素与蛋白质和各种酶混合时可发生强烈的反应,从而使得蛋白质和酶失去活性。当重金属元素积累到一定浓度,会导致急性或亚急性中毒或慢性中毒等。食物中重金属元素的含量超标事件时有发生,对消费者的健康产生了潜在的威胁,因此检测食品中重金属的含量十分重要[55-56]。
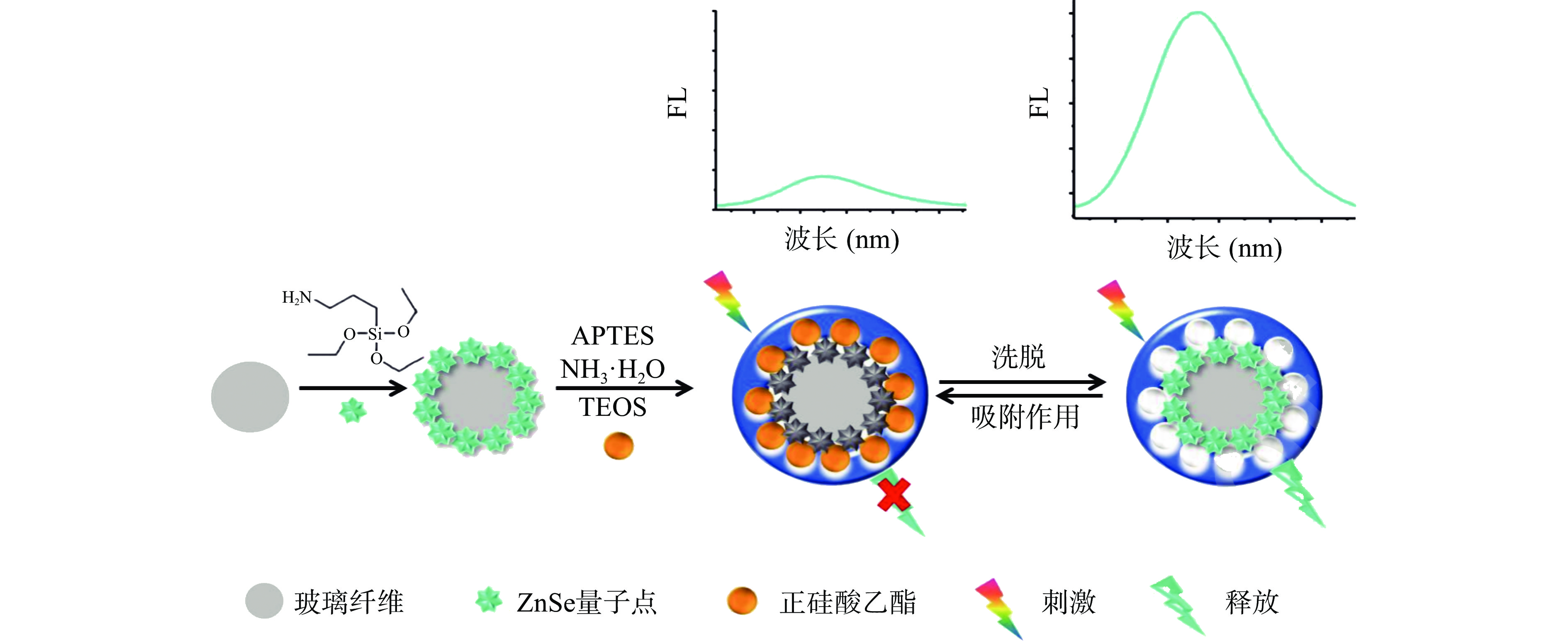
食品中的重金属污染物包括汞、铜、铅和镉等。Qi等[57]提出了一种基于离子印迹聚合物的新型三维微流控纸基分析装置,用于同时检测Cu2+和Hg2+。Cu2+在0.11~58 µg/L之间具有良好的线性关系,LOD为0.035 µg/L,Hg2+线性范围为0.26~34 µg/L,LOD为0.056 µg/L。该装置检测Cu2+和Hg2+具有良好的选择性、灵敏度和特异性。同时,因其可同时检测两种离子,节省检测时间,为将来开发多离子检测器件奠定了基础。Zhou等[58]在三维旋转微流控纸芯片平台上实现了一种基于离子印迹技术实现对Cd2+和Pb2+的特异性检测(图5)。该技术采用硒化锌量子点进行检测,与碲化镉量子点相比,硒化锌量子点的毒性更小,更有利于环境保护。Cd2+在1~70 µg/L之间具有良好的线性度,LOD为0.245 µg/L,Pb2+在1~60 µg/L范围内呈线性变化,LOD为0.335 µg/L。该技术够实现Cd2+和Pb2+的特异性和多通道检测且对两种离子的检验均具有灵敏性。三维微流控纸基分析装置的研发,使纸基器件从平面走向立体,同时新设计的三维旋转µPADs展示的定量检测信息,为未来快速检测目标金属离子提供了新的研究方向。
2.4 食品添加剂检测
添加食品添加剂已经成为增强或改变食物颜色或味道的常规做法,已广泛应用于食品工业中。食品添加剂若使用不规范可对人体健康造成潜在的危害[59-60]。因此,开发高灵敏及可靠的分析方法用于食品添加剂的检测非常关键。
亚硝酸盐是食品添加剂和腐蚀抑制剂的常见离子,可以与胺相互作用形成致癌的亚硝胺化合物[61]。Rajasulochana等[62]描述了一个用于定量检测亚硝酸盐的三维微流控纸基传感器,该3D打印平台可重复使用且可以同时为测试多个样品提供支持,并利用智能手机进行数字图像采集和比色分析。亚硝酸盐浓度在0.1~10 ppm的范围内呈线性变化,LOD和LOQ分别为0.12 ppm和0.35 ppm,相对标准偏差为1.31%,即具有良好的再现性。该传感器已成功应用于实际水样中亚硝酸根的测定,回收率在83.5%~109%之间。基于微流控纸基传感器所构建的检测方法,由于具有简单、易于制造和使用等优势,在食品分析检测中具有广阔的应用前景。莱克多巴胺(Ractopamine,RAC)是一种β促效剂药物,是瘦肉精的一种。Kordasht等[63]成功研制了一种新型纸基电化学适体传感器,用于RAC的检测。使用设计的三电极纸基生物装置系统对RAC的浓度进行评估。在0.001 µmol/L~100 mmol/L范围内测定RAC,定量下限为0.01 µmol/L,测定结果具有良好的灵敏度和选择性。适体在电化学纸基传感器中的开发将能够增强对其检测原理的理解,为开发低成本、便携、敏感和选择性纸基传感器奠定基础。
食用色素可以提升食品色泽,增加人的食欲,已成为食品中最常见的添加剂之一。Wu等[64]通过智能手机成像在纸基分析设备上对多种分析物进行灵敏、快速的比色检测。以饮料中常见的食用色素亮蓝和苋菜红为检测目标,通过色素在纸基设备上的堆叠富集和分离,利用梯度区电泳增强分离效果,采用智能手机进行实时成像。得到亮蓝和苋菜红的LOD分别为0.35和0.14 μg/mL。这种方法具有同时分离和富集样品的功能,检测结果与分光光度法检测结果接近,在进行多组分分析时也可避免光谱干扰,具有简单、快捷和灵敏等优势,为开发低成本、便携式和高灵敏度的检测器件奠定了基础。
3. 结论与展望
近年来,纸基微流控技术因其成本低廉、反应灵敏、非专业人员可使用和快速获得检测结果等优势,被科研人员大量研究,广泛应用于食品分析和生化分析等多个领域。利用生物分子、纳米材料和聚合物材料制成的分子探针被用于μPADs中,提高了μPADs的特异性和灵敏度。分子探针材料对μPADs的表面功能化,能够以低成本、快速高效的方式传达准确的分析结果。信息技术的高速发展,物联网、云计算和微流体等技术的融合促进了μPADs与智能手机的应用软件的开发,使智能手机能够应用于现场快速检测。然而,在实现电子分析方法的优化之前,还需要克服多学科的障碍。μPADs的开发应用弥补了现存方法依赖实验室大型仪器的不足。但是作为一种新型的快速检测技术,其研究还是停留在实验室阶段,缺乏市场化应用,纸张润湿条件不明及样品在纸张上的挥发等问题还有待解决。人工智能的发明,可以促进μPADs自动进样的研究,使其早日实现自动化进样,提高检测效率。
-
表 1 纸基微流控不同传感材料性能对比研究
Table 1 Performance comparison of different sensing materials for paper-based microfluidics
探针材料 检测方法 检测物质 LOD 参考文献 格里斯试剂 纸芯片比色法 亚硝酸盐 15 µmol/L [17] 间苯二酚与7-硝基苯并呋喃嗪 纸芯片荧光法 半胱氨酸 16 nmol/L [19] ZnFe2O4磁性纳米粒子 纸芯片比色传感器 双酚A 6.18 nmol/L [28] 用抗体和辣根过氧化物酶修饰的纳米金-单链DNA 纸芯片比色传感器 病原体抗原 10 pg/mL [29] 溴甲酚绿试剂 智能手机三维纸芯片比色法 白蛋白 3.68 g/L [30] 氯酚红-β-D-半乳吡喃糖苷和磷酸品红 纸芯片比色法 金黄色葡萄球菌和大肠杆菌 106 CFU/mL [31] 2′-羟基查尔酮衍生物 纸芯片荧光法 白蛋白 16.4 nmol/L [32] 以二乙烯基砜作为同型二功能试剂的碳点 纸芯片荧光法 生物硫醇 0.3 μmol/L [26] 1,5-二苯基碳酰二肼修饰的纳米银 纸芯片比色法 半胱氨酸和同型半胱氨酸 0.16 μmol/L [33] 金纳米粒子适配体 智能手机纸芯片比色法 多氯联苯 70 ng/L [34] 碘化汞 纸芯片比色法 重金属汞 20 mg/L [35] -
[1] ZHOU X, PU H, SUN D W. DNA functionalized metal and metal oxide nanoparticles: Principles and recent advances in food safety detection[J]. Critical Reviews in Food Science and Nutrition,2021,61(14):2277−2296. doi: 10.1080/10408398.2020.1809343
[2] CHEONG Y H, GE L, LISAK G. Highly reproducible solid contact ion selective electrodes: Emerging opportunities for potentiometry-a review[J]. Analytica Chimica Acta,2021,1162:338304. doi: 10.1016/j.aca.2021.338304
[3] EGGHE T, VAN GUYSE J F R, GHOBEIRA R, et al. Evaluation of cross-linking and degradation processes occurring at polymer surfaces upon plasma activation via size-exclusion chromatography[J]. Polymer Degradation and Stability,2021,187:109543. doi: 10.1016/j.polymdegradstab.2021.109543
[4] GREER B, CHEVALLIER O, QUINN B, et al. Redefining dilute and shoot: The evolution of the technique and its application in the analysis of foods and biological matrices by liquid chromatography mass spectrometry[J]. TrAC Trends in Analytical Chemistry,2021,141:116284. doi: 10.1016/j.trac.2021.116284
[5] CHEN T, YIN S, WU J. Nanomaterials meet microfluidics: Improved analytical methods and high-throughput synthetic approaches[J]. TrAC Trends in Analytical Chemistry,2021,142:116309. doi: 10.1016/j.trac.2021.116309
[6] LIU X R, HUANG Y F, HUANG J J. Identification of benzophenone analogs in rice cereal through fast pesticide extraction and ultrahigh-performance liquid chromatography-tandem mass spectrometry[J]. Foods,2022,11(4):572−584. doi: 10.3390/foods11040572
[7] CHEN G, LIU G, JIA H, et al. A sensitive bio-barcode immunoassay based on bimetallic Au@Pt nanozyme for detection of organophosphate pesticides in various agro-products[J]. Food Chemistry,2021,362:130118. doi: 10.1016/j.foodchem.2021.130118
[8] RONG Y, ALI S, OUYANG Q, et al. Development of a bimodal sensor based on upconversion nanoparticles and surface-enhanced Raman for the sensitive determination of dibutyl phthalate in food[J]. Journal of Food Composition and Analysis,2021,100:103929. doi: 10.1016/j.jfca.2021.103929
[9] KARIMI-MALEH H, KARIMI F, REZAPOUR M, et al. Carbon paste modified electrode as powerful sensor approach determination of food contaminants, drug ingredients, and environmental pollutants: A review[J]. Current Analytical Chemistry,2019,15(4):410−422. doi: 10.2174/1573411014666181026100037
[10] YU Y, YANG Z, WANG L, et al. LAMP for the rapid diagnosis of iridovirus in aquaculture[J]. Aquaculture and Fisheries,2022,7(2):158−165. doi: 10.1016/j.aaf.2021.08.002
[11] JEONG S G, KIM D H, KIM J, et al. Programmable microfluidic flow for automatic multistep digital assay in a single-sheet 3-dimensional paper-based microfluidic device[J]. Chemical Engineering Journal,2021,411:128429. doi: 10.1016/j.cej.2021.128429
[12] SELVAKUMAR B, KATHIRAVAN A. Sensory materials for microfluidic paper based analytical devices-A review[J]. Talanta,2021,235:122733. doi: 10.1016/j.talanta.2021.122733
[13] XI J, FENG J, GE D, et al. Preparation of carbon/Al2O3/nZVI magnetic nanophase materials produced from drinking water sludge for the removal of As (V) from aqueous solutions[J]. Environmental Science and Pollution Research,2021,28(6):7261−7270. doi: 10.1007/s11356-020-11084-5
[14] NGUYEN H A, LEE N Y. Polydopamine aggregation: A novel strategy for power-free readout of loop-mediated isothermal amplification integrated into a paper device for multiplex pathogens detection[J]. Biosensors and Bioelectronics,2021,189:113353. doi: 10.1016/j.bios.2021.113353
[15] LIU X, WU W, CUI D, et al. Functional micro-/nanomaterials for multiplexed biodetection[J]. Advanced Materials,2021,33(30):2004734−2004783. doi: 10.1002/adma.202004734
[16] PRAPAPORN S, ARISARA S, WUNPEN C, et al. Nanocellulose films to improve the performance of distance-based glucose detection in paper-based microfluidic devices[J]. Analytical Sciences,2020,36(12):1447−1452. doi: 10.2116/analsci.20P168
[17] ARVAND M, ARJMANDI N, SHAKIBAIE M, et al. Colorimetric microfluidic paper-based sensor for determination of nitrite in drinking water with enhanced color development[J]. Journal of Physics D:Applied Physics,2020,53(35):355403−355419. doi: 10.1088/1361-6463/ab8e7a
[18] ZHOU J, YU C, LI Z, et al. A rapid and highly selective paper-based device for high-throughput detection of cysteine with red fluorescence emission and a large Stokes shift[J]. Analytical Methods,2019,11(10):1312−1316. doi: 10.1039/C9AY00041K
[19] ZHANG J, MIAO Y, CHENG Z, et al. A paper-based colorimetric assay system for sensitive cysteine detection using a fluorescent probe[J]. Analyst,2020,145(5):1878−1884. doi: 10.1039/C9AN02271F
[20] PARINAZ A, MOHAMMAD H, FARZAD S, et al. An innovative colorimetric platform for the low-cost and selective identification of Cu (II), Fe (III), and Hg (II) using GQDs-DPA supported amino acids by microfluidic paper-based (µPADs) device: Multicolor plasmonic patterns[J]. Journal of Environmental Chemical Engineering,2021,9(5):106197−106213. doi: 10.1016/j.jece.2021.106197
[21] FENG L X, TANG C, HAN X X, et al. Simultaneous and sensitive detection of multiple small biological molecules by microfluidic paper-based analytical device integrated with zinc oxide nanorods[J]. Talanta,2021,232:122499. doi: 10.1016/j.talanta.2021.122499
[22] JIANG H, GUO Q, ZHANG C, et al. Microfluidic origami nano-aptasensor for peanut allergen Ara h1 detection[J]. Food Chemistry,2021,365:130511. doi: 10.1016/j.foodchem.2021.130511
[23] SHARIATI S, KHAYATIAN G. Microfluidic paper-based analytical device using gold nanoparticles modified with N, N′-bis (2-hydroxyethyl) dithiooxamide for detection of Hg (ii) in air, fish and water samples[J]. New Journal of Chemistry,2020,44(43):18662−18667. doi: 10.1039/D0NJ03986A
[24] PINHEIRO T, MARQUES A C, CARVALHO P, et al. Paper microfluidics and tailored gold nanoparticles for nonenzymatic, colorimetric multiplex biomarker detection[J]. ACS Applied Materials & Interfaces,2021,13(3):3576−3590.
[25] SAWETWONG P, CHAIRAM S, JARUJAMRUS P, et al. Enhanced selectivity and sensitivity for colorimetric determination of glyphosate using Mn-ZnS quantum dot embedded molecularly imprinted polymers combined with a 3D-microfluidic paper-based analytical device[J]. Talanta,2021,225:122077. doi: 10.1016/j.talanta.2020.122077
[26] ORTIZ-GOMEZ I, ORTEGA-MUÑOZ M, MARÍN-SÁNCHEZ A, et al. A vinyl sulfone clicked carbon dot-engineered microfluidic paper-based analytical device for fluorometric determination of biothiols[J]. Microchimica Acta,2020,187(7):1−11.
[27] SHIBATA H, HIRUTA Y, CITTERIO D. Fully inkjet-printed distance-based paper microfluidic devices for colorimetric calcium determination using ion-selective optodes[J]. Analyst,2019,144(4):1178−1186. doi: 10.1039/C8AN02146E
[28] KONG Q, WANG Y, ZHANG L, et al. A novel microfluidic paper-based colorimetric sensor based on molecularly imprinted polymer membranes for highly selective and sensitive detection of bisphenol A[J]. Sensors and Actuators B:Chemical,2017,243:130−136. doi: 10.1016/j.snb.2016.11.146
[29] HUANG J Y, LIN H T, CHEN T H, et al. Signal amplified gold nanoparticles for cancer diagnosis on paper-based analytical devices[J]. ACS sensors,2018,3(1):174−182. doi: 10.1021/acssensors.7b00823
[30] YANG R J, TSENG C C, JU W J, et al. Integrated microfluidic paper-based system for determination of whole blood albumin[J]. Sensors and Actuators B:Chemical,2018,273:1091−1097. doi: 10.1016/j.snb.2018.07.010
[31] ASIF M, AWAN F R, KHAN Q M, et al. based analytical devices for colorimetric detection of S. aureus and E. coli and their antibiotic resistant strains in milk[J]. Analyst,2020,145(22):7320−7329. doi: 10.1039/D0AN01075H
[32] LUO Z, LV T, ZHU K, et al. Paper-based ratiometric fluorescence analytical devices towards point-of-care testing of human serum albumin[J]. Angewandte Chemie,2020,132(8):3155−3160. doi: 10.1002/ange.201915046
[33] SHARIATI S, KHAYATIAN G. The colorimetric and microfluidic paper-based detection of cysteine and homocysteine using 1, 5-diphenylcarbazide-capped silver nanoparticles[J]. RSC Advances,2021,11(6):3295−3303. doi: 10.1039/D0RA08615K
[34] ZHANG D, WANG Y, LI C, et al. Polychlorinated biphenyl detection in organic solvents with paper-based analytical devices[J]. Environmental Technology,2021,42(11):1766−1771. doi: 10.1080/09593330.2019.1680741
[35] NASHUKHA H L, SITANURAK J, SULISTYARTI H, et al. Simple and equipment-free paper-based device for determination of mercury in contaminated soil[J]. Molecules,2021,26(7):2004−2021. doi: 10.3390/molecules26072004
[36] QIN G, NIU Z, YU J, et al. Soil heavy metal pollution and food safety in China: Effects, sources and removing technology[J]. Chemosphere,2021,267:129205. doi: 10.1016/j.chemosphere.2020.129205
[37] NORDHAGEN S, LAMBERTINI E, DEWAAL C S, et al. Integrating nutrition and food safety in food systems policy and programming[J]. Global Food Security,2022,32:100593. doi: 10.1016/j.gfs.2021.100593
[38] HUA Z, YU T, LIU D, et al. Recent advances in gold nanoparticles-based biosensors for food safety detection[J]. Biosensors and Bioelectronics,2021,179:113076. doi: 10.1016/j.bios.2021.113076
[39] PAUK V, KREJČÍ M, LEMR K. Unified chromatography-mass spectrometry as a versatile tool for determination of food dyes[J]. Analytica Chimica Acta,2021,1157:338401. doi: 10.1016/j.aca.2021.338401
[40] SHIN D, KIM J, KANG H S. Simultaneous determination of multi-pesticide residues in fish and shrimp using dispersive-solid phase extraction with liquid chromatography-tandem mass spectrometry[J]. Food Control,2021,120:107552. doi: 10.1016/j.foodcont.2020.107552
[41] LIANG Z, ABDELSHAFY A M, LUO Z, et al. Occurrence, detection, and dissipation of pesticide residue in plant-derived foodstuff: A state-of-the-art review[J]. Food Chemistry,2022,384:132494. doi: 10.1016/j.foodchem.2022.132494
[42] KIM T H, HAHN Y K, KIM M S. Recent advances of fluid manipulation technologies in microfluidic paper-based analytical devices (µPADs) toward multi-step assays[J]. Micromachines,2020,11(3):269−299. doi: 10.3390/mi11030269
[43] ROMERO-GONZÁLEZ R. Detection of residual pesticides in foods[J]. Foods,2021,10(5):1113−1116. doi: 10.3390/foods10051113
[44] WU L, ZHOU M, LIU C, et al. Double-enzymes-mediated Fe2+/Fe3+ conversion as magnetic relaxation switch for pesticide residues sensing[J]. Journal of Hazardous Materials,2021,403:123619. doi: 10.1016/j.jhazmat.2020.123619
[45] OROUJI A, ABBASI-MOAYED S, HORMOZI-NEZHAD M R. ThThnated development of a pH assisted AgNP-based colorimetric sensor array for simultaneous identification of phosalone and azinphosmethyl pesticides[J]. Spectrochimica Acta Part A:Molecular and Biomolecular Spectroscopy,2019,219:496−503. doi: 10.1016/j.saa.2019.04.074
[46] DENG S, YANG T, ZHANG W, et al. Rapid detection of trichlorfon residues by a microfluidic paper-based phosphorus-detection chip (µPPC)[J]. New Journal of Chemistry,2019,43(19):7194−7197. doi: 10.1039/C9NJ00898E
[47] NSIBANDE S A, FORBES P B C. Development of a quantum dot molecularly imprinted polymer sensor for fluorescence detection of atrazine[J]. Luminescence,2019,34(5):480−488. doi: 10.1002/bio.3620
[48] YANG N, ZHOU X, YU D, et al. Pesticide residues identification by impedance time-sequence spectrum of enzyme inhibition on multilayer paper-based microfluidic chip[J]. Journal of Food Process Engineering,2020,43(12):13544−13555.
[49] LIU R, ALI S, HARUNA S A, et al. Development of a fluorescence sensing platform for specific and sensitive detection of pathogenic bacteria in food samples[J]. Food Control,2022,131:108419. doi: 10.1016/j.foodcont.2021.108419
[50] GUPTA R, RAZA N, BHARDWAJ S K, et al. Advances in nanomaterial-based electrochemical biosensors for the detection of microbial toxins, pathogenic bacteria in food matrices[J]. Journal of Hazardous Materials,2021,401:123379. doi: 10.1016/j.jhazmat.2020.123379
[51] WANG J, LEI Y, YU Y, et al. Use of acetic acid to partially replace lactic acid for decontamination against Escherichia coli O157: H7 in fresh produce and mechanism of action[J]. Foods,2021,10(10):2406−2420. doi: 10.3390/foods10102406
[52] KARUPPIAH S, MISHRA N C, TSAI W C, et al. Ultrasensitive and low-cost paper-based graphene oxide nanobiosensor for monitoring water-borne bacterial contamination[J]. ACS Sensors,2021,6(9):3214−3223. doi: 10.1021/acssensors.1c00851
[53] PUIU M, BALA C. Microfluidics-integrated biosensing platforms as emergency tools for on-site field detection of foodborne pathogens[J]. TRAC Trends in Analytical Chemistry,2020,125:115831. doi: 10.1016/j.trac.2020.115831
[54] PARK C, LEE J, KIM Y, et al. 3D-printed microfluidic magnetic preconcentrator for the detection of bacterial pathogen using an ATP luminometer and antibody-conjugated magnetic nanoparticles[J]. Journal of Microbiological Methods,2017,132:128−133. doi: 10.1016/j.mimet.2016.12.001
[55] MUKHERJEE S, BHATTACHARYYA S, GHOSH K, et al. Sensory development for heavy metal detection: A review on translation from conventional analysis to field-portable sensor[J]. Trends in Food Science & Technology,2021,109:674−689.
[56] NG H K M, LIM G K, LEO C P. Comparison between hydrothermal and microwave-assisted synthesis of carbon dots from biowaste and chemical for heavy metal detection: A review[J]. Microchemical Journal,2021,165:106116. doi: 10.1016/j.microc.2021.106116
[57] QI J, LI B, WANG X, et al. Three-dimensional paper-based microfluidic chip device for multiplexed fluorescence detection of Cu2+ and Hg2+ ions based on ion imprinting technology[J]. Sensors and Actuators B: Chemical,2017,251:224−233. doi: 10.1016/j.snb.2017.05.052
[58] ZHOU J, LI B, QI A, et al. ZnSe quantum dot based ion imprinting technology for fluorescence detecting cadmium and lead ions on a three-dimensional rotary paper-based microfluidic chip[J]. Sensors and Actuators B:Chemical,2020,305:127462. doi: 10.1016/j.snb.2019.127462
[59] EVSTATIEV R, CERVENKA A, AUSTERLITZ T, et al. The food additive EDTA aggravates colitis and colon carcinogenesis in mouse models[J]. Scientific Reports,2021,11(1):1−12. doi: 10.1038/s41598-020-79139-8
[60] COX S, SANDALL A, SMITH L, et al. Food additive emulsifiers: A review of their role in foods, legislation and classifications, presence in food supply, dietary exposure, and safety assessment[J]. Nutrition Reviews,2021,79(6):726−741. doi: 10.1093/nutrit/nuaa038
[61] FLORES M, TOLDRÁ F. Chemistry, safety, and regulatory considerations in the use of nitrite and nitrate from natural origin in meat products-invited review[J]. Meat Science,2021,171:108272. doi: 10.1016/j.meatsci.2020.108272
[62] RAJASULOCHANA P, GANESAN Y, KUMAR P S, et al. Based microfluidic colorimetric sensor on a 3D printed support for quantitative detection of nitrite in aquatic environments[J]. Environmental Research,2022,208:112745. doi: 10.1016/j.envres.2022.112745
[63] KORDASHT H K, SAADATI A, HASANZADEH M. A flexible paper based electrochemical portable biosensor towards recognition of ractopamine as animal feed additive: Low cost diagnostic tool towards food analysis using aptasensor technology[J]. Food Chemistry,2022,373:131411. doi: 10.1016/j.foodchem.2021.131411
[64] WU Z Y, ZHANG F, KUANG Z, et al. Fast and sensitive colorimetric detection of pigments from beverages by gradient zone electrophoresis on a paper based analytical device[J]. Microchemical Journal,2022,179:107499. doi: 10.1016/j.microc.2022.107499





 下载:
下载:
 下载:
下载:
